Sie befinden sich hier
Inhalt
TGF-Beta signaling in chronic liver disease development and progression
We analyze the role of TGF-β signaling in chronic liver disease, e.g. alcohol dependent fibrosis, cirrhosis and hepatocellular carcinoma (HCC). One focus of our research is plasticity and differentiation of liver cells (hepatocytes). We investigate epithelial-mesenchymal transition of hepatocytes (EMT) during liver disease progression by cell biology, molecular und systems biology approaches such as quantitative immunohistochemistry, RT-PCR, Western Blotting. We also analyze the switch of tumor suppressive TGF-β signaltransduction in early stages of disease towards tumor promotive effects in late HCC using cell lines, human tissue and selected transgene mouse models. Our work is supported by different DFG grants and is embedded into the Virtual Liver Network (BMBF) as well as the IT Liver Consortium (Marie Curie ITN, EU).
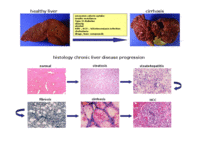
The significance of chronic liver injury
Alcohol induced chronic liver damage is the 10th most frequent cause of death of male in Germany. 2 % of all deaths in our country in 2005, i.e. 16.806 persons, are due to chronic liver damage (Statistisches Bundesamt Wiesbaden). Alcoholic liver injury usually develops from fatty liver degeneration called steatosis. Ongoing alcohol abuse or other negative influences induce the development of fibrosis.
Further histopathological changes include liver cirrhosis, often followed by liver failure and/or the development of liver cancer. Although many diagnostical and therapeutical methods for are available to date, liver transplantation is the only possible treatment of final liver failure.
Upon chronic damage, e.g., by hepatitis virus infection or alcohol abuse, the wound healing response of the liver derails and excessive deposition of ECM leads to organ fibrosis with increasing loss of liver function. The wound healing process is initiated by a complex network of cytokines, including pro-fibrotic proteins TGF-β and CTGF.
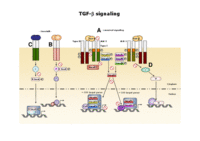
TGF-Beta signaling
TGF-β signaling is required for activation of HSCs, which is considered the most important event in development of fibrosis. Therefore,TGF-β is sometimes termed "master cytokine" of liver fibrogenesis. TGF-β signals through transmembrane receptors that stimulate cytoplasmic Smad proteins, which modulate transcription of target genes, including those encoding ECM, e.g. procollagen-I and –III.
By functional criteria, Smad proteins are subdivided into three classes: receptor Smads (R-Smad), common mediator Smads (Co-Smads) and inhibitory Smads (I-Smads). Smad7, an I-Smad, blocks R-Smad phosphorylation and subsequent downstream events by forming a stable complex with the activated TGF-β type I receptor.
Inhibitory Smad7 is very efficient to blunt TGF-β effects in general and was previously used by us and others to intervene with fibrogenesis in chronic diseases of liver, kidney, lung and skin.
Signaling pathways described are activated during liver damage and wound healing. Upon ongoing chronic injury, e.g. by alcohol abuse or hepatitis B or C virus infection, these signaling pathways are running excessively and induce fibrogenic mechanisms with different cell types in the liver involved.
Hepatocytes in liver fibrogenesis – new insights
One main focus of our studies is now to clarify the role of hepatocytes in the fibrotic liver response. Starting out from in vitro experiments with primary hepatocyte cultures and immortalized AML-12 cells, TGF-β signaling was assessed and apoptosis was found to be a minor effect. Instead hepatocytes undergo EMT, a physiologic process in embryogenesis and of relevance for cancerous cell transformation. In injured liver however this process contributes to the promotion of fibrosis. Already after a few days of culture, hepatocytes lose their epithelial honey-comb-like shape towards a fibroblast like phenotype. Further we could demonstrate by microarray analysis, that stimulation of hepatocytes with TGF-β regulates the expression of genes involved in cell cycle, inflammation, metabolism, apoptosis, EMT, and fibrosis. We confirmed regulation of several known targets, e.g., GADD45β, Bim and Bcl-2. However, we also found Snail, a known mediator of EMT, induced. Connective Tissue Growth Factor (CTGF), a strong inducer of fibrosis, was also detected. In a mouse model, hepatocyte-specific overexpression of Smad7 was achieved. Thereby a fibrogenic response after CCl4 intoxication could be blunted. These results emphasize the dynamic nature of liver fibrosis, challenge the paradigm of HSC as crucial source for liver myofibroblasts and hint at a prominent role for hepatocytes in liver fibrogenesis.
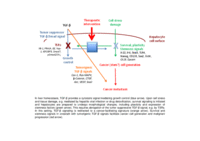
TGF-Beta signaltransduction in HCC
Sustained TGF-β signaling activation during regeneration drives progression of chronic liver disease by promoting fibrogenesis and suppressing hepatocyte proliferation. While TGF-β has cytostatic and thus tumor suppressive effects, i.e. apoptosis activation and inhibition of proliferation, on hepatocytes in liver homeostasis, liver regeneration and early stages of liver disease, this effect is switched towards tumor promotion in late stage disease and hepatocellular carcinoma. During hepatocarcinogenesis cytostatic TGF-β effects are abrogated which facilitates cancer stem cell (CSC) formation and hepatocyte dedifferentiation and promotes epithelial mesenchymal transition (EMT) and metastasis. Thus, efficacy of a TGF-β-directed therapy is context dependent throughout the disease process necessitating specified approaches based on molecular and pathomorphological data.
As the tumor suppressive to tumor promoting switch of TGF-β signaling is not sufficiently delineated yet, we expect additional relevant TGF-β signaling regulators (TSRs). Identification and molecular characterization of TSRs is central aim of our work significantly improve HCC diagnosis, prognosis and therapeutical options in the future.
Kontextspalte
Contact person

Prof. Dr. rer. nat. Steven Dooley
Molecular Hepatology
Phone +49 621/383-3768
Fax +49 621/383-1467
PhD- or Masterthesis
If you are interested in performing your Master or PhD/MD in our group, please contact Prof. Dr. rer. nat. Steven Dooley or go to
https://doktorboerse.fimm-online.de/index.php?action=show&inst=7
or
Publications
Publications of this group can be found on pubmed.
Grants
- BMBF (LiSyM, GoBio)
- DFG
- CSC – Chinese Scholarship Council
- Chinesisch-Deutsches Zentrum für Wissenschaftsförderung