Vascular Signaling, Metabolism and Cancer
Head: Prof. Andreas Fischer
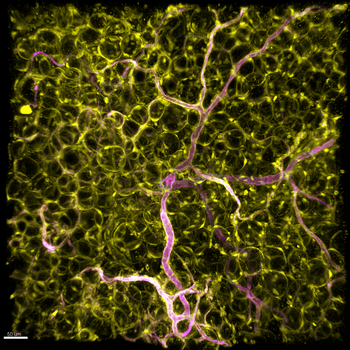
Blood vessels deliver oxygen, nutrients, and hormones to every organ. The endothelium—the inner lining of all vessels—forms one of the body’s largest barrier surfaces and serves as a gateway for metabolite and hormone exchange. Positioned at the frontline of systemic sensing, endothelial cells regulate traffic across the vessel wall and secrete angiocrine factors that influence organ function.
Our mission is to understand how endothelial cells respond to physiological and pathophysiological changes. We combine cutting-edge cellular and molecular biology with in vivo models to unravel how angiocrine signaling shapes organ function and homeostasis.